In what Thermo Fisher Scientific officials are calling an “historic, path-breaking event,” the FDA has granted premarket approval to the company’s Oncomine Dx Target Test, which screens tumor samples for biomarkers associated with three FDA-approved drugs for non-small cell lung cancer (NSCLC).
The panel-based test evaluates 23 different biomarkers simultaneously and can be used to identify patients who may be eligible for treatment with three distinct therapies: the combined therapy of Tafinlar and Mekinist, Xalkori, or Iressa.
“By now making the clear claim that the same panel can be used to look at multiple drugs with one test, it moves away from the sequential testing of cancer to simultaneous testing,” said Joydeep Goswami president of clinical NGS and oncology at Thermo Fisher. “That is really important. Take lung cancer, which (this first panel tests for). More than 85% of patients are diagnosed at Stage 3, the patients are very sick and you want to get them to the right treatment the first time.”
By having the ability to test for multiple drugs simultaneously, the Oncomine Dx Target Test helps take the guesswork out of sequential testing, whereby clinicians act on a hunch to test for one particular drug that may benefit the patient. If the result from a single-drug companion diagnostic comes back negative after several weeks, it can significantly delay getting the patient the right drug based on their cancer’s profile.
“It is time wasted; and during that time, the patient is getting ever sicker,” Goswami added.
Simultaneous with the premarket approval for the diagnostic, the FDA also approved the combination therapy for NSCLC of Taflinar and Mekinist for patients with BRAF V600E mutation as detected by the new test. The Oncomine Dx Target Test also detects the presence of BRAF, ROS1, and EGFR gene mutations or alterations in tumor tissue of patients with NSCLC.
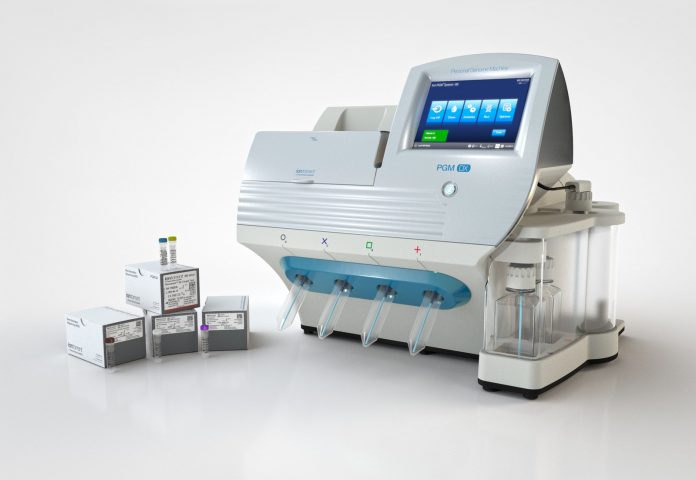
All tests will be run on Thermo Fisher's Ion PGM Dx System, which received FDA 510(k) clearance in parallel for use on formalin-fixed, paraffin-embedded (FFPE) tissue samples. LabCorp's Diagnostics and Covance businesses, NeoGenomics Laboratories and Cancer Genetics, are the first laboratories to will offer the Oncomine Dx Target Test.
The premarket approval comes a little more than a year and a half after Thermo Fisher struck a deal with both Novartis and Pfizer to develop the diagnostic. At the time, both companies had robust NSCLC pipelines that included four drugs in various stages of development. Pfizer’s drug Xalcori had already received regulatory approval at that time.
“The potential to generate a paradigm shift through this agreement—from one test for one drug, to one test for multiple NSCLC therapies—represents a significant step forward in realizing the promise of precision medicine,” noted Thermo Fisher’s Mark Stevenson, president of life sciences solutions upon entering the collaboration.
According to Goswami, the test and the Ion PGM Dx System on which it is run are especially well suited for NSCLC because they require a small quantity of DNA to deliver and answer. “All of these diagnoses today are based on a tissue biopsy. For a lung cancer patient, it is often quite hard to get a tissue biopsy, and the small amounts you get are precious,” he said.
More broadly, Goswami noted the new test marks a significant breakthrough for NGS technology and its application as an approved tool for diagnostics. “It is a big deal in terms of validating NGS as a IVD product, which means this can now be adopted in any CLIA- and CAP-certified lab across the country, and other geographies where we are filing this,” he concluded. “It democratizes the availability of NGS.”
Tafinlar and Mekinist are registered trademarks of Novartis; Xalkori is a registered trademark of Pfizer; and Iressa is a registered trademark of AstraZeneca.