University of Cambridge researchers have grown ‘mini brains’ in the lab that model amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD), two conditions that often overlap, and have also successfully tested a candidate drug on them.
While such mini brains have been grown before, they have often been short lived in the past making it difficult to use them to study neurodegenerative diseases like ALS. For the first time, this study showed it is possible to grow them for almost a year.
New and more effective treatments for neurodegenerative diseases such as ALS/FTD are desperately needed. The team hopes the insights they can gain from their model could help develop better therapeutics for these devastating conditions.
Lab grown ‘organoids’ are being developed to model various different organs such as the liver, heart and brain, and more accurately test reactions of human tissues to exposure with a variety of different drugs than using animals or cell lines alone. While the first versions of these mini organs were fairly basic, they are becoming more sophisticated and realistic by the day as the technology and knowledge of the area improves.
These organoids are usually created by extracting cells from a patient’s skin, reprogramming these cells back to the stem cell stage and then differentiating them into whichever cell and organ type is required. This allows researchers to create specific models for certain diseases, particularly if they are due to some kind of genetic mutation carried by the patient.
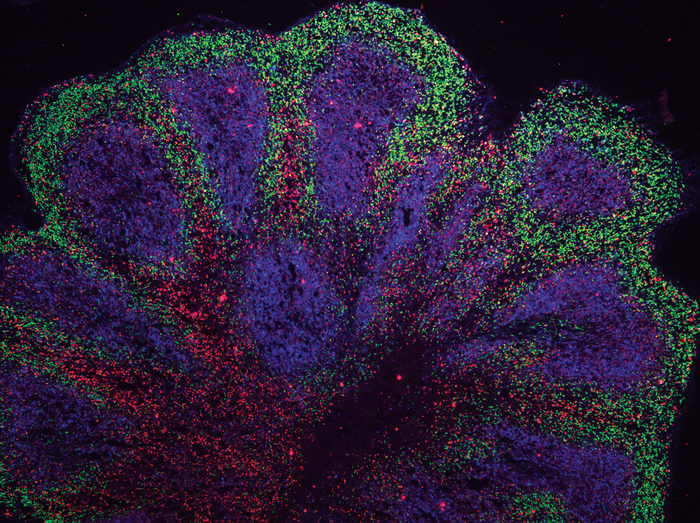
In this study, which is published in Nature Neuroscience, scientists based at the John van Geest Centre for Brain Repair, University of Cambridge, extracted cells from patients suffering from overlapping ALS/FTD (C9ORF72 mutation) to grow brain organoids for 240 days – in unpublished work the same group has managed to grow the mini brains for 340 days.
A key difference from other similar organoids that other groups have grown was that these were grown as brain ‘slices’ rather than a whole standard shaped mini brain, which helped to make sure all the cells received a good level of nutrition.
During the growth of the organoids the team was able to observe subtle signs indicating abnormalities seen in ALS/FTD. Using single cell sequencing and a range of different assays, they observed proteomic and DNA repair disruption in both astroglial cells and neurons.
More specifically, the astroglia had increased levels of the autophagy signaling protein P62 and some of the neurons had high levels of the dipeptide repeat protein poly(GA) and showed DNA damage and early signs of cell death.
Notably, the team managed to reverse many of these changes using a drug candidate known as GSK2606414, this included toxic protein buildup, cell stress and nerve cell loss. Drugs similar to this candidate are now being tested in clinical trials seeking to find treatments for neurodegenerative disease.
“Neurodegenerative diseases are very complex disorders that can affect many different cell types and how these cells interact at different times as the diseases progress. To come close to capturing this complexity, we need models that are more long-lived and replicate the composition of those human brain cell populations in which disturbances typically occur, and this is what our approach offers,” said András Lakatos, the senior author who led the research based in Cambridge’s Department of Clinical Neurosciences, in a press statement.
“Not only can we see what may happen early on in the disease – long before a patient might experience any symptoms – but we can also begin to see how the disturbances change over time in each cell.”