Remdesivir, a nucleotide analogue that inhibits viral RNA polymerases, is emerging as an early leader in the quest for antiviral medications against SARS-CoV-2, the virus causing COVID-19. Results from a clinical study with compassionate-use remdesivir showed that clinical improvement was observed in 36 of 53 patients (68%). Overall mortality in treated patients was 13%, lower than in previously published studies of non-treated COVID-19 patients with serious symptoms. This trial’s findings, funded by the drug’s manufacturer Gilead Sciences, were published in the New England Journal of Medicine.
Remdesivir has shown activity against several viruses, including the Ebola virus and coronaviruses like the original SARS virus and Middle East respiratory syndrome (MERS) coronavirus. And, in vitro studies indicate activity against SARS-CoV-2. Based on remdesivir’s activity profile and the fact that it appeared to be well-tolerated in Ebola patients receiving the drug in past virus outbreaks, researchers were eager to test it in the current COVID-19 outbreak.
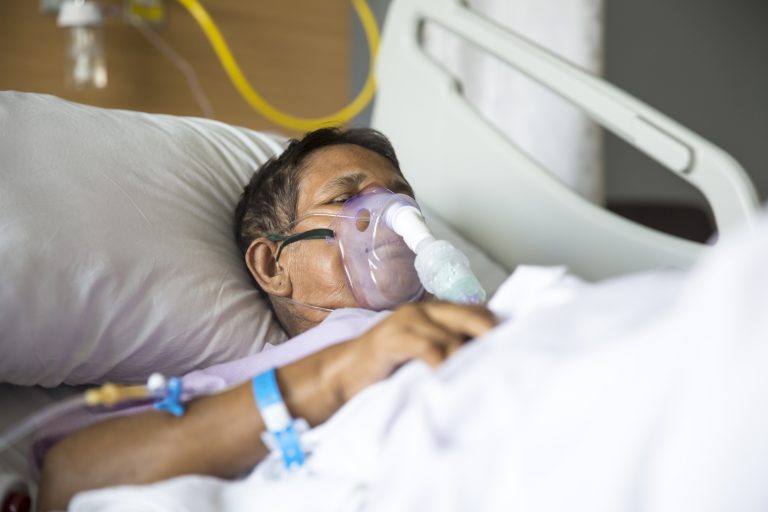
The study included 61 hospitalized patients with COVID-19 with an oxygen saturation of 94 percent or less while breathing on their own or with oxygen support. The study’s analysis included data from 53 patients as data from 8 could not be analyzed; 22 were in the United States, 22 in Europe or Canada, and 9 in Japan. Notably, a majority of the study participants were severely ill. At the start of the study, 30 patients (57%) were receiving mechanical ventilation and 4 (8%) were receiving extracorporeal membrane oxygenation, or ECMO.
Of the 53 patients included in the analysis, 40 (75%) received a full 10-day course of remdesivir, 10 (19%) received 5 to 9 days of treatment, and 3 (6%) fewer than 5 days of treatment.
At 18 days post-treatment, about two-thirds of all patients improved—36 of 53 patients (68%)— based on the level of oxygen support needed. It is especially notable that 17 of 30 patients (57%) who were receiving mechanical ventilation were extubated, and 3 of 4 patients (75%) receiving ECMO stopped receiving it.
Further, at the conclusion of the study follow-up, 25 of 53 patients (47%) were discharged, and 7 patients (13%) died. Of those who 7 patients who died, mortality was 18% (6 of 34) among patients receiving invasive ventilation and 5% (1 of 19) among those not receiving invasive ventilation.
The lack of a control group in the study complicates what role the drug had in producing the improvements. Still, the lower than anticipated mortality rate of 13% is encouraging. In other COVID-19 studies, mortality rates range widely from 17% to 78% of patients receiving mechanical ventilation. This study group included a majority of patients with severe symptoms of disease as evidence by the need for mechanical ventilation or ECMO. In addition, 75% of patients were male, most were over 60 years of age and had coexisting conditions – all known risk factors for poorer outcomes with COVID-19.
This compassionate-use study had notable limits: small patient numbers, less than three weeks of follow up, missing or lack of data on 8 patients initially treated, and most importantly, the lack of a placebo-controlled, randomized study design. Still, this analysis does suggest that remdesivir may have clinical benefit in patients with severe COVID-19.
Remdesivir is currently in at least six clinical trials in moderate and severe COVID-19. Included in these are two Phase III randomized, open-label, multicenter clinical studies in countries with high prevalence of COVID-19.